Co2 molecular geometry9/10/2023  
The double bonds between oxygen-carbon and sulfur-carbon as well as the equal number of lone pairs of electrons on both sulfur and oxygen atoms exert an almost similar force of repulsion to give a symmetrical structure to the carbonyl sulfide molecule. The bond length between oxygen-carbon is 115.78 pm and sulfur-carbon is 156.01pm. Moreover, the molecular geometry of this molecule can be studied in detail with the help of the Valence Shell Electron Pair Repulsion (VSEPR) Theory. Molecular Geometry of Carbonyl Sulfide (OCS)įrom the Lewis structure, it is clear that the molecular geometry of carbonyl sulfide is linear as all three participating elements are arranged at 180° from one another. Otherwise, the central atom will not share its valence electrons and the formation of a new molecule will not take place. Low electronegativity is essential for the central atom as it only then will share most of its valence electrons. It is so because the lower the electronegativity value, the higher will be the tendency of sharing the electrons. Why must the central atom in a molecule have the lowest electronegativity? 
Hence, sharing of valence electrons takes place among all the participating atoms to achieve a stable condition.īoth single bond and triple bond shares the odd number of valence electrons where its only double bond with which the molecule has achieved a stable condition. As carbonyl sulfide is a covalent compound, donation of valence electrons is not possible. It is clear from the structure that both oxygen and sulfur atoms need two valence electrons to complete their octet. 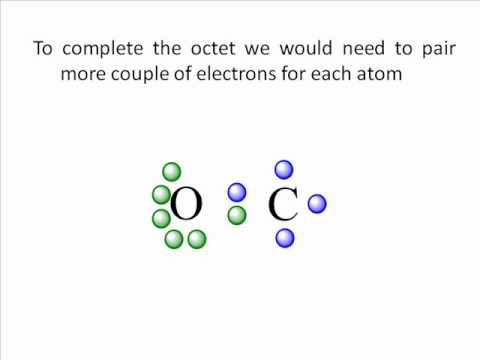
Why are double bonds formed in carbonyl sulfide? Step 6: Collate all the aforementioned points and draw the Lewis structure of carbonyl sulfide: Step 5: Search for the central atom: It will be carbon as it has the lowest electronegativity value out of all the three participating atoms. Step 4: Determine the bond formation taking place between the participating atoms: Double bond will form as oxygen and sulfur will bond with carbon only for two valence electrons each. Step 3: Determine the number of valence electrons further needed to stabilize one carbonyl sulfide molecule: It is 8 as the total number of valence electrons required is 24. Step 2: Determine the total number of valence electrons available to draw the Lewis structure of carbonyl sulfide: It is 16 for one OCS molecule. Step 1: Determine the valence electrons for each participating atom: It is four for carbon and six for both oxygen as well as sulfur. Steps to Draw the Lewis structure of carbonyl sulfide Sulfur has 6 valence electrons and the atomic number 16. Oxygen has six valence electrons and atomic number eight. To begin with the Lewis structure of carbonyl sulfide, first, we need to study the same for all the participating elements.Ĭarbon has four valence electrons and the atomic number six. The maximum number of valence electrons an atom can have is eight. Due to this, electrons in the outermost shell do not feel the nucleus’s force of attraction and readily bond with another element available nearby. The nucleus exerts a force of attraction which helps electrons with showcasing their natural properties without jumping in other orbits.īy this logic, the farther the orbit is weaker will be the force of attraction by the nucleus. The explanation of this lies in the structure of an atom where the nucleus is present at the center and electrons revolve around it in their orbits. The valence electrons are present in the outermost shell of the atom and participate in the bond formation. The Lewis structure is drawn by keeping the symbol of an element at the center and drawing valence electrons in pairs around it. Conclusion Lewis Structure of Carbonyl Sulfide (COS) 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
